Our Research
Understanding the pathologies of acoustic trauma
To model mechanical damage to the inner ear, we have developed a technique to generate a strong wave stimulus to produce mechanical injury in zebrafish lateral line organs. Overstimulated lateral-line organs show morphological and functional changes that correspond to changes observed in noise-exposed mammalian ears. Yet lateral-line hair cells and nerve connections begin to repair hours after exposures and show full morphological and functional recovery within days. Understanding how zebrafish hair cells are damaged then repaired following mechanical injury may ultimately reveal potential therapeutics for treating sensorineural hearing loss. This project is funded by a Research Project Grant from the National Institutes of Health (NIH/NIDCD).
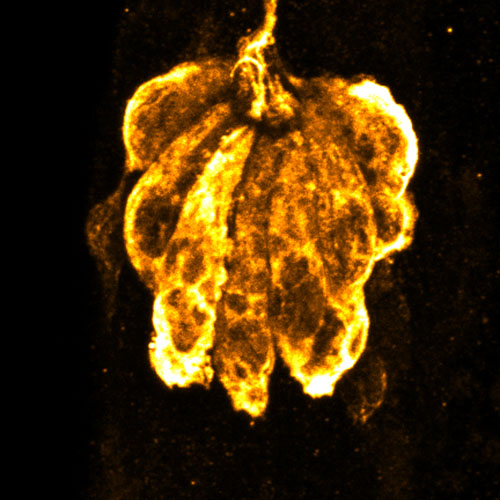
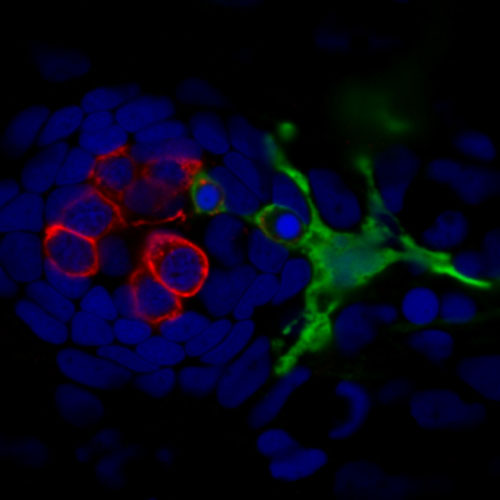
Identifying methods to protect from cisplatin-induced damage
Cisplatin is an effective and widely used drug used to treat many forms of cancer. But cisplatin is associated with toxic side effects, including neuropathy and permanent hearing loss. In collaboration with Dr. Mark Warchol, we have optimized a zebrafish model ototoxicity and neurotoxicity with the goal of uncovering therapeutic strategies to protect sensory hair cells and nerve fibers from damage during cisplatin-based chemotherapy. This project is funded by The Children’s Discovery Institute, St Louis Children’s Hospital.
Defining the effects of lateral-line organ damage on zebrafish behavior
The zebrafish lateral line is a well-established model for investigating the cellular mechanisms of hair-cell organ damage. Yet the associations between lateral line damage and changes in biologically relevant behavior are not well defined. Lateral line organs sense water flow and contribute to a behavior called positive rheotaxis-orienting and swimming against oncoming water flow. Using a custom micro flume to generate a calibrated water flow stimuli, we record zebrafish behavior with a high-speed camera, then quantify fish position and movement using DeepLabCut, a 3D marker less pose estimation software that uses deep neural networks and machine learning. With this data we can annotate and classify zebrafish rheotaxis to determine how damage from mechanical damage or toxic drugs affects lateral-line mediated behavior.
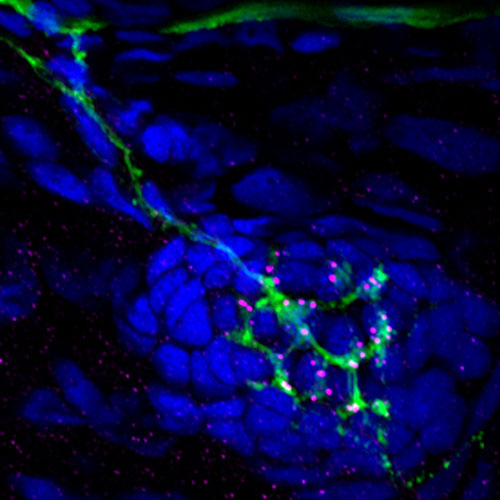
Identifying novel regulators of afferent nerve regeneration and hair-cell reinnervation
In contrast to mammalian inner ears, zebrafish ears and lateral-line organs have the remarkable ability to repair and regenerate lost hair cells and neurons. In a project funded by RNID (previously known as Action on Hearing Loss) and the McDonnell Center for Cellular Neuroscience, we are striving to identify molecular pathways to promote cochlear nerve regeneration and hair cell reinnervation.